Free Introduction to Macromolecules OCR A Level Biology revision notes – covering specification points 2.1.2(b) and 2.1.2(c).
Want to go deeper?
Macromolecules
Macromolecules are large biomolecules, important to the structure and function of living organisms, made up of covalently bonded monomers.
The table below gives an overview of these four groups of biomolecules.
| Biomolecule | Elements Present | Monomer | Polymer(s) | Examples |
|---|---|---|---|---|
| Carbohydrates | C, H, O | Monosaccharides | Polysaccharides | Maltose, Sucrose, Lactose, Starch, Glycogen, Cellulose |
| Lipids | C, H, O | Fatty acids & Glycerol | Triglycerides, Phospholipids |
Phospholipids, Cholesterol, Steroids |
| Proteins | C, H, O, N, S* *most |
Amino acids | Polypeptides, Proteins |
Collagen, Enzymes, Antibodies |
| Nucleic Acids | C, H, O, N, P | Nucleotides | DNA, RNA | mRNA, tRNA, rRNA |
Monomers are small molecules which, when joined to other molecules of the same type, form a polymer.
A polymer is a large molecule made from many monomers joined together in condensation reactions.
A condensation reaction is when two molecules are joined together with a covalent bond, forming (and releasing) a water molecule in the process.
The diagram below shows a covalent bond (a glycosidic bond) being formed from a reaction between the hydroxyl groups on two different molecules:
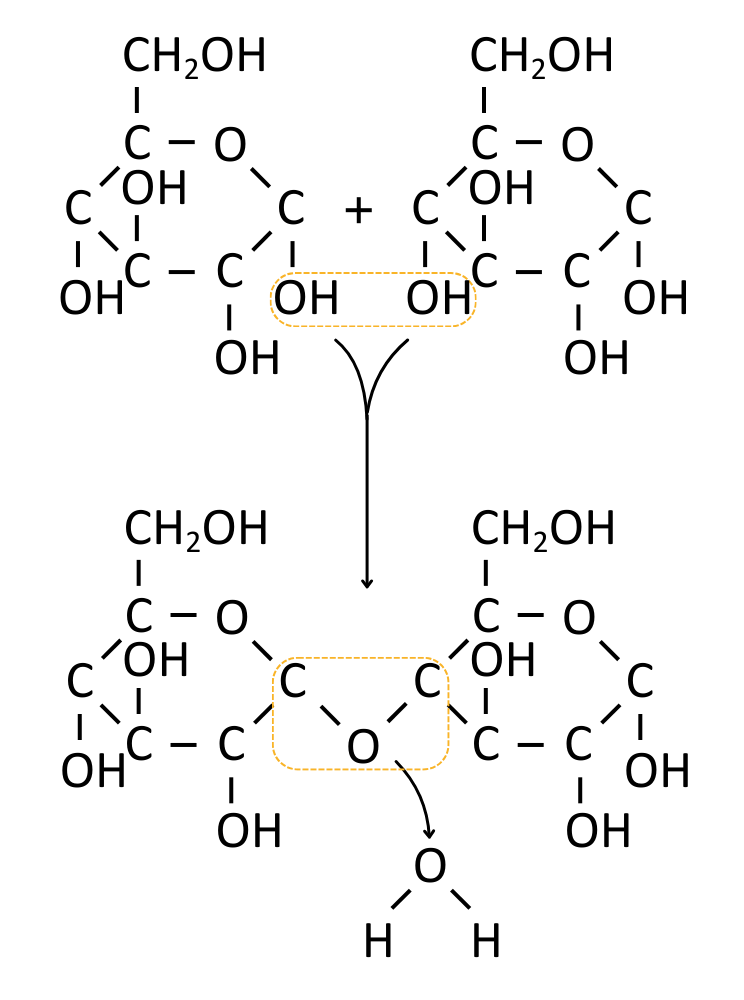
The table below gives an overview of the bonds in biological molecules formed by condensation reactions.
| Biomolecule | Monomer | Bond Type | Image |
|---|---|---|---|
| Carbohydrates | Monosaccharides | Glycosidic |

|
| Proteins | Amino acids | Peptide |

|
| Lipids | Glycerol & Fatty acids | Ester |
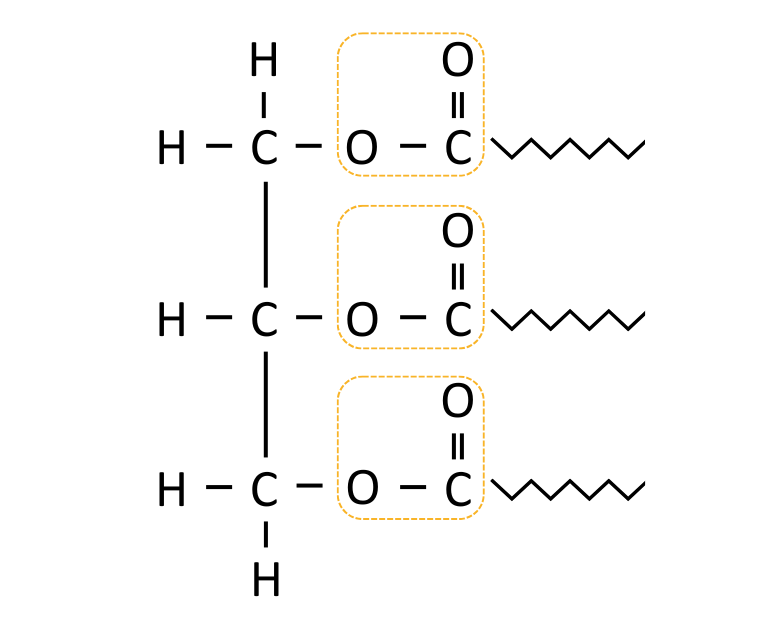
|
| Nucleic Acids | Nucleotides | Phosphodiester |
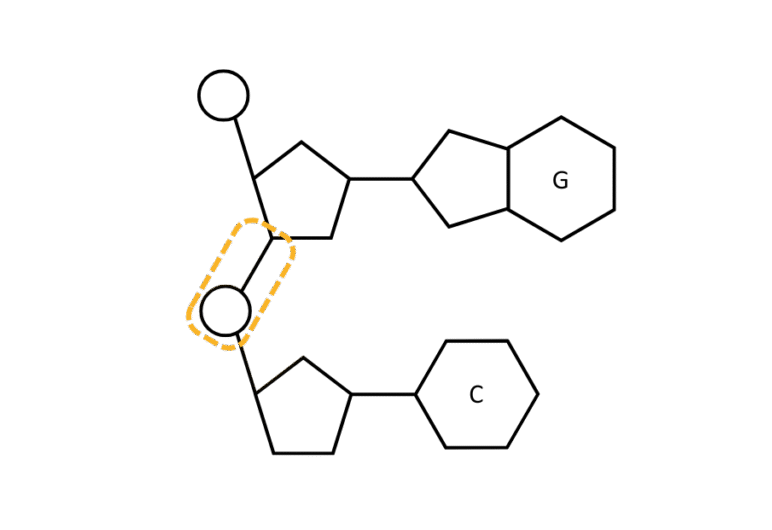
|
Polymers can be broken down (digested) into monomers again in hydrolysis reactions.
A hydrolysis reaction is when a water molecule is used to break a covalent bond, producing two molecules from one.
The diagram below shows a covalent bond (specifically a peptide bond) being broken in a dipeptide to produce two amino acids.

