Free The Importance of Excretion revision notes for OCR A Level Biology – covering specification point 5.1.2 (a).
Excretion
Excretion is the removal of metabolic waste from the body.
Metabolic waste is the unwanted and potentially harmful byproducts of cellular metabolism.
If metabolic byproducts can accumulate to significant levels, then reactions can slow down as enzyme activity is affected, or cellular damage can occur if they are toxic.
The table below outlines some examples of the harmful impacts of metabolic waste on cellular activity:
| Harmful Effect | Metabolic Waste | Mechanism |
|---|---|---|
| Enzyme activity decreases as changes in pH disrupt their tertiary structure. | Carbon dioxide (CO2). | Forms carbonic acid and releases H+ ions, lowering pH. |
| Enzymes and structural proteins denature, halting metabolic reactions. | Ammonia (NH3). | Highly toxic and alkaline; interferes with hydrogen bonding. |
| Oxygen transport efficiency falls, reducing aerobic respiration rates. | Carbon dioxide (CO2). | Reduces haemoglobin’s affinity for oxygen. |
| Cells lose water by osmosis, leading to dehydration and reduced metabolic efficiency. | Urea / Excess ions. | Lowers tissue water potential, causing water loss from cells. |
The Importance of Excretion
Excretion is essential for maintaining homeostasis within an organism.
Because metabolic by-products are continuously produced by cellular reactions, they must be removed efficiently to avoid them accumulating.
The table below outlines some examples of metabolic wastes:
| Metabolic Waste | Excretory Organ | Excretory Importance |
|---|---|---|
| Carbon dioxide (CO2) | Lungs | Excess CO2 forms carbonic acid, lowering pH and reducing haemoglobin’s oxygen affinity. |
| Ammonia (NH3) | Liver → Kidneys | Highly toxic; converted to urea for safe excretion. |
| Urea | Kidneys | Nitrogenous waste is removed in urine; it controls osmotic balance. |
Importance of Excreting Carbon Dioxide
Aerobically respiring cells produce CO₂, which diffuses into the bloodstream and forms carbonic acid (H₂CO₃).
Carbonic acid can then dissociate into hydrogencarbonate ions and hydrogen ions (H⁺ + HCO₃⁻).
Most carbonic acid dissociates inside red blood cells (being catalysed by carbonic anhydrase), but some dissociation still occurs (slowly) in the blood plasma.
The process is outlined below:
CO₂ + H₂O ⇌ H₂CO₃ ⇌ H⁺ + HCO₃⁻
Small increases in the hydrogen ion concentration of the blood plasma are detected by the respiratory centre in the medulla oblongata of the brain (as a change in blood plasma pH), which increases the breathing rate to remove more carbon dioxide.
The diagram below shows the position of the medulla oblongata:
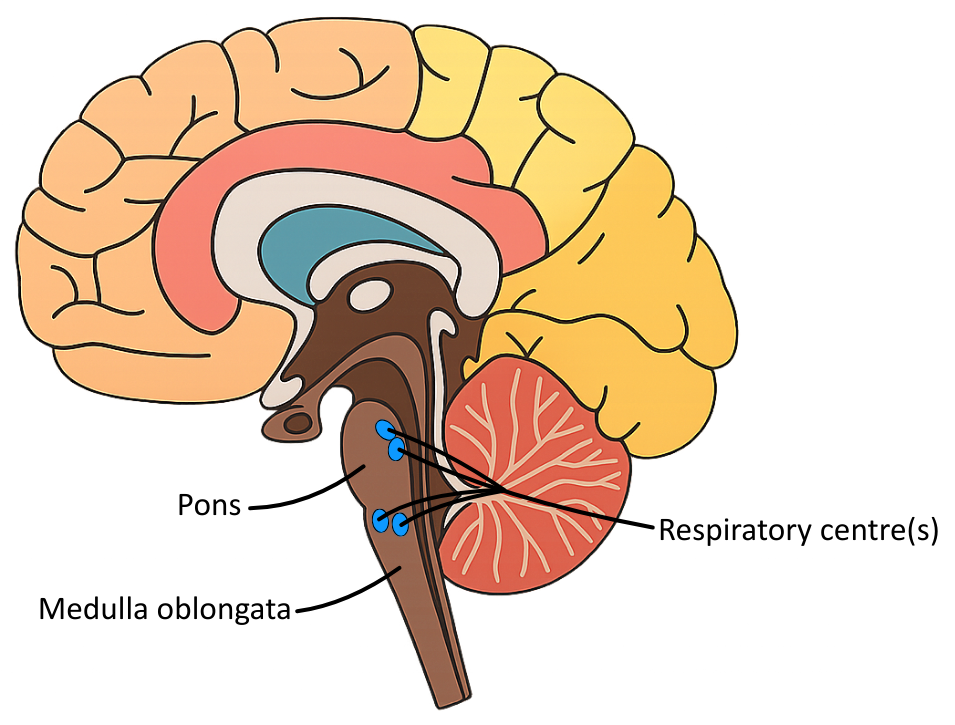
It is useful to know that the respiratory centre is not a centralised area, and is instead several clusters of neurones dispersed across the medulla oblongata and the pons; you are not expected to know this.
Importance of Excreting Nitrogenous Waste: Ammonia/Urea
The digestion of proteins produces amino acids, which are useful for protein synthesis but, in excess, cannot be stored for later use. Because amino acids are also a source of energy, excreting them directly would be wasteful.
The liver metabolises amino acids, removing the amine group (NH₂) in deamination to produce a keto acid and ammonia (NH3).
The process is outlined below:
Amino acid + O₂ → Keto acid + NH₃
Keto acids can be used in respiration or converted into a carbohydrate or fat for storage.
The ammonia (NH3) formed from the amine group (NH2) is highly soluble and toxic* and must be converted into a safer and less soluble compound before it accumulates: urea.
Urea, made by the liver, can circulate more safely in the blood before being filtered from the blood plasma by the kidneys and excreted in urine.
*It is useful to know that, whilst the amine group is commonly described as toxic, it is itself not toxic, and will quickly form ammonia (toxic) in solution, or bind to an organic compound (not in itself toxic).
Importance of Excreting Water
Although water is essential for hydration and as a solvent for biochemical reactions, water in excess causes dilution of the blood and tissue fluids. This decreases the solute concentration of the blood plasma, disrupting osmotic balance and affecting enzyme activity.
The kidneys are the main organs involved in regulating water levels. They filter the blood and reabsorb the appropriate amount of water to maintain a stable blood water potential.
Osmoregulation is coordinated by the hypothalamus, which detects changes in blood osmotic potential using osmoreceptors and alters the activity of the kidney accordingly.




